Small packages: UI nanotechnology firm gets $2 million boost in fight against cancer
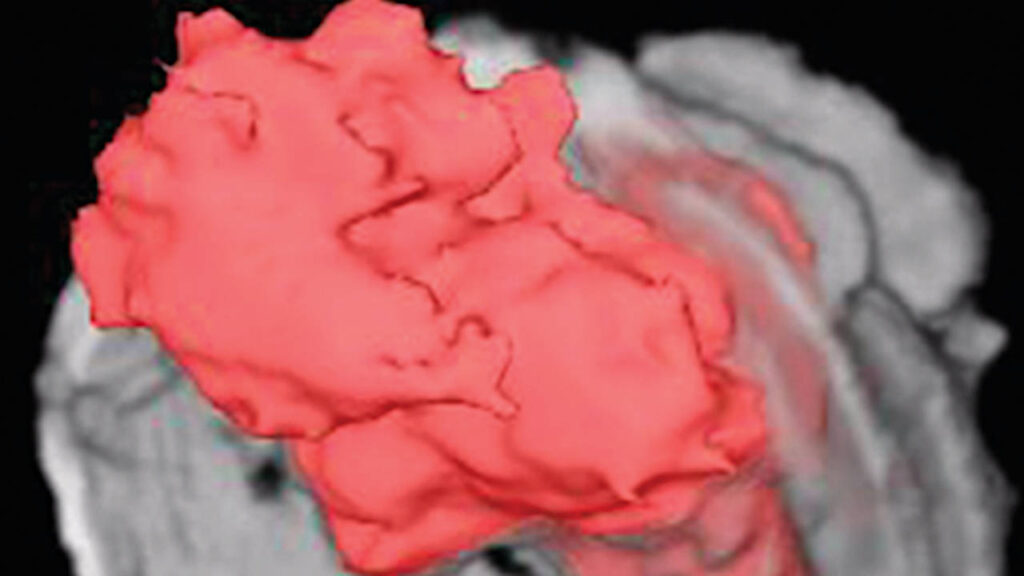
Coralville-based NanoMedTrix is using nanoparticles to deliver chemotherapy drugs directly to cancerous growths. The tiny particles also include a contrast agent to allow tumors to be visualized, as in this 3D virtual cystoscopy.
A novel technology that delivers anti-cancer drugs via nanoparticles to tumors – and helps physicians monitor them in real time — has won a University of Iowa spin-out a $2 million Phase II Small Business Innovation Research (SBIR) grant from the National Cancer Institute to help the company find a partner in the pharmaceutical world.
NanoMedTrix, founded by Jose Assouline, an adjunct associate professor in the Roy J. Carver Department…

Want to Read More?
Get immediate, unlimited access to all subscriber content and much more.
Learn more in our subscriber FAQ.
Do you want to read and share this article without a paywall?







